Atavistic Oncology: A New Dawn in Cancer Therapy
Atavistic Oncology, conceived in 2011 by Dr. Frank Arguello, represents a groundbreaking shift in cancer treatment and scientific understanding.
Following the publication of Dr. Arguello’s influential book, “Atavistic Metamorphosis: A New and Logical Explanation on the Origin and Biological Nature of Cancer Cells,” atavistic oncology has emerged as a leading approach in the fight against cancer.
This innovative field offers a new perspective on the true origin and biological behavior of cancer cells, leading to the development of atavistic chemotherapy.
Further, Atavistic Oncology Cancer treatment utilizes a unique combination of FDA-approved drugs to target and eliminate cancer cells selectively.
Atavistic chemotherapy stands out for its effectiveness and non-toxic nature, offering a home-based treatment option under the guidance of an oncologist or family doctor.
Unlike alternative cancer treatments, atavistic chemotherapy provides a scientifically validated method to eradicate primary, metastatic, and inoperable/unresectable cancers.
Clinical Trial Expectations
Patients eligible for the Atavistic Chemotherapy Clinical Trial can expect to see a significant clinical and/or radiological response within the first three weeks, with the potential for complete eradication of cancer within six months, or they owe nothing.
Don't Waste Your Life!
The Truth About Conventional Cancer Treatments
Our Patients' Cancers Before and After Atavistic Chemotherapy
(Click Images to Enlarge)
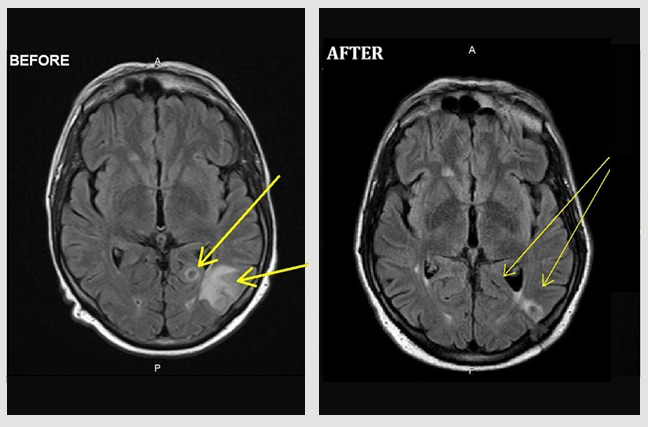
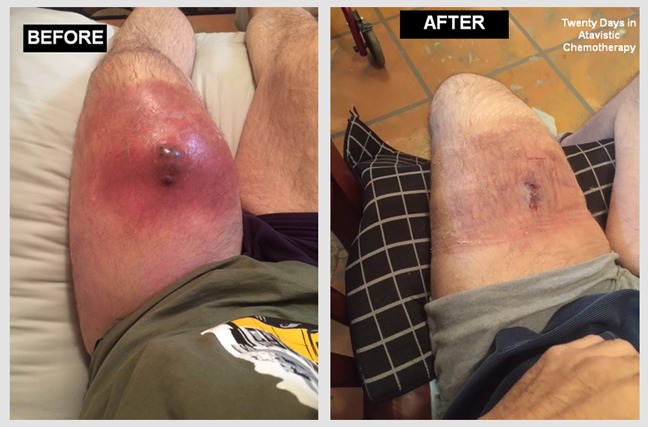
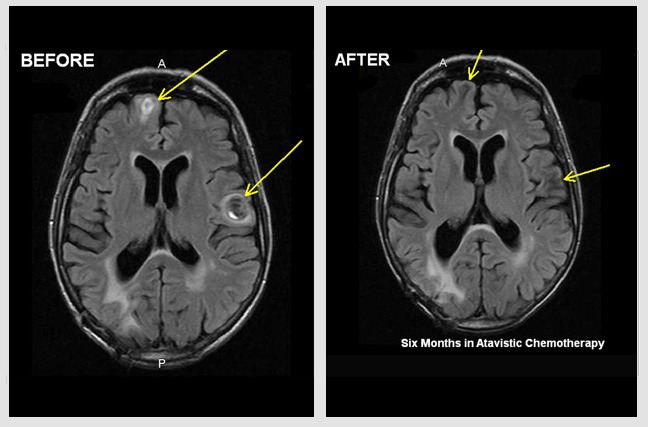
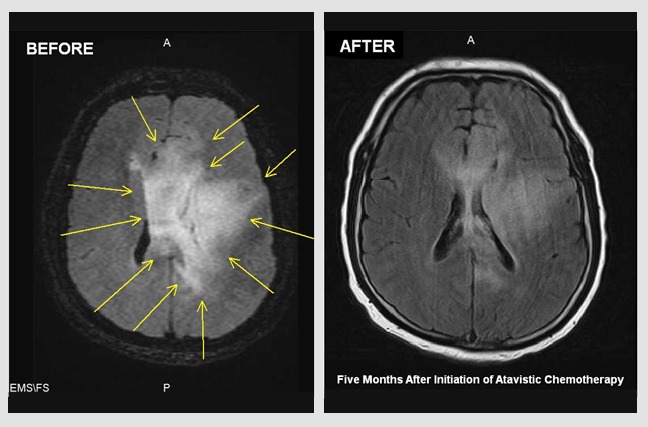
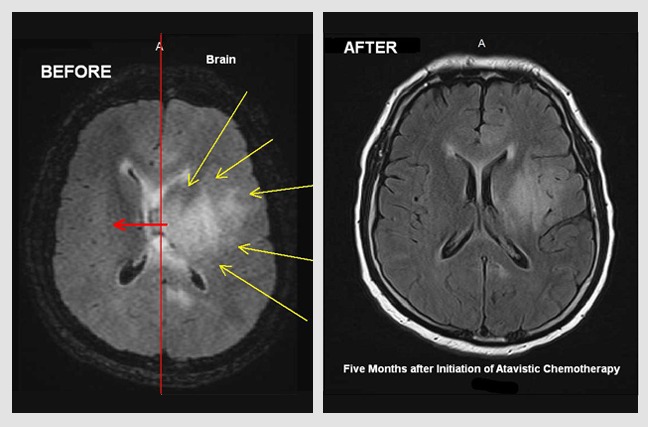
Primary Diffuse B-Cell, EBV-Associated Lymphoma of the Brain.
Male (54 Y/O) had been under immunosuppression for the last 9 years following a lung transplant. In late November 2019, the patient experienced seizures and was rushed to the hospital. An MRI of head demonstrated a diffuse brain lesion (yellow arrows in images). Following a needle biopsy, the lesion was diagnosed as a primary B-Cell, EBV-Associated Lymphoma. The ‘before’ image is from an MRI of November 23, 2019, prior to initiation of atavistic chemotherapy. The ‘after’ image is an MRI twenty days of initiated atavistic chemotherapy.
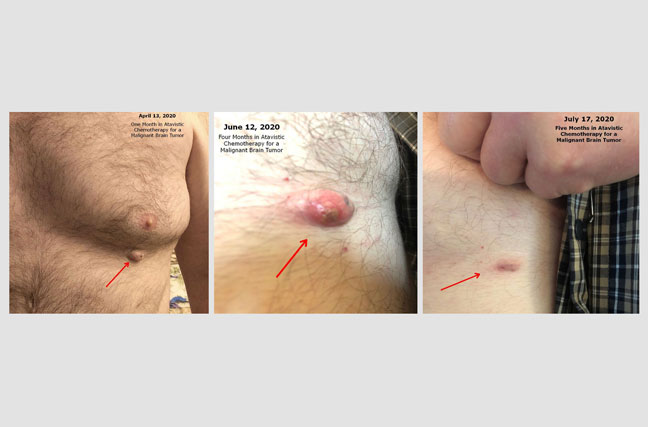
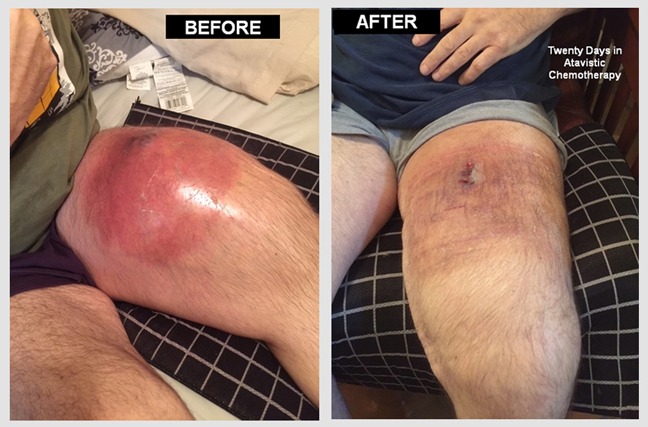
Regression of Benign Lipoma During Atavistic Chemotherapy for a Malignant Brain Tumor:
Randy is a 45 Y/O patient who suffers from a Grade III Anaplastic Oligodendroglioma (a malignant brain tumor partially resected surgically in February 2020). Although the “Before and After” images related to the brain tumor results will be posted here soon, we wanted to show you an unexpected event. For years, the patient had had a slow growing mass below her left breast which was diagnosed clinically as a lipoma (a benign tumor formed by fat cells or adipocytes). During treatment with atavistic chemotherapy for his brain tumor, the patient noticed changes in his benign lipoma. He recorded these changes with photographs, starting after the first month of treatment. As you could see, the tumor underwent complete regression by the fifth month in atavistic chemotherapy. Atavistic chemotherapy may be helpful also in benign tumors. The reduction of body hair is due to the treatment.
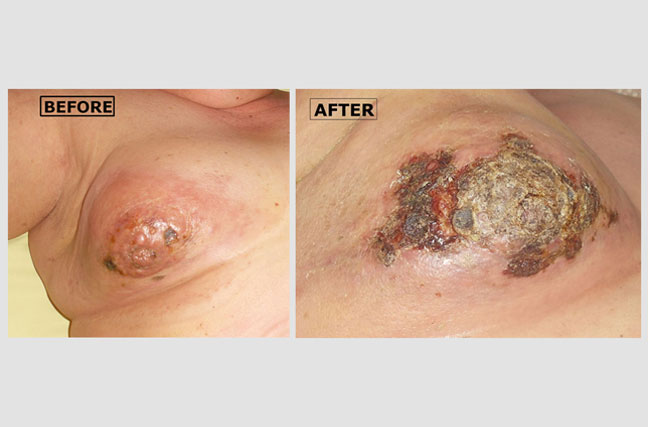
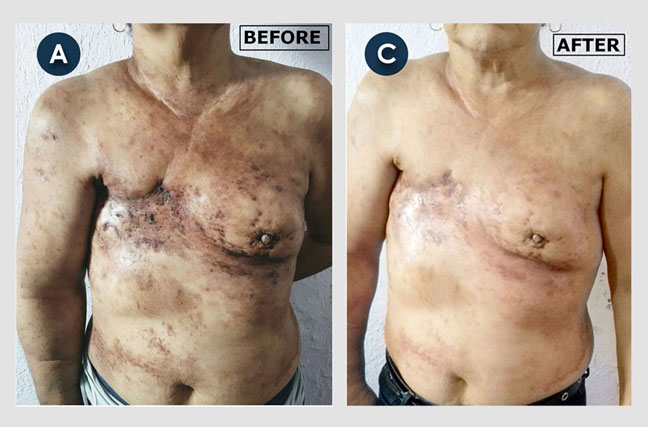
Inflammatory Breast Cancer (part 1):
Angelina, a 58-year-old woman came to us with aggressive breast cancer. It had extensively invaded the skin and areola (the area around her nipple) of her right breast. The 'before' picture shows multiple raised lumps of cancerous tissue growing under the skin of her breast and areola. Most of her breast skin looks reddish because more blood is flowing to the area. The 'after' photo shows lots of necrotic (dead) cancerous tissue that had formed by four weeks after beginning atavistic chemotherapy.
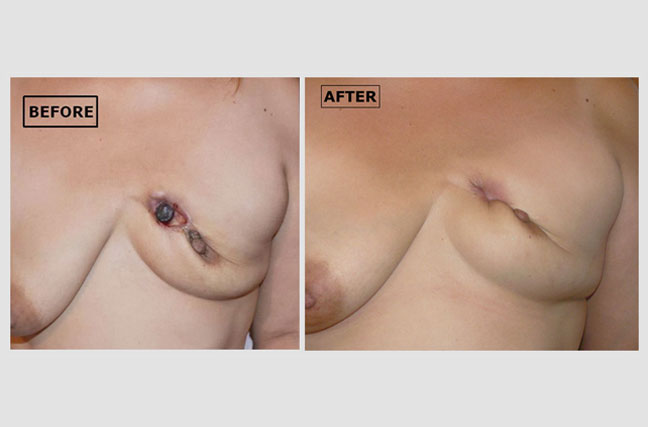
Inflammatory Breast Cancer (part 2):
Angelina, the patient in the previous case study, continued her therapy with us. The 'before' picture shows a well-healing scab where the dead cancer tissue is beginning to separate from the healthy tissue. The ‘after’ photo shows the result: a healing wound in her right breast after the scab has come away. The picture was taken after completing atavistic chemotherapy.
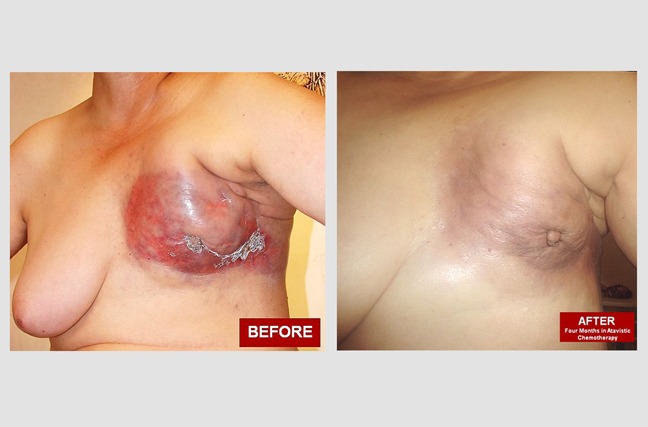
Locally Advanced Breast Cancer:
Bertha, a 46-year-old woman, had decided against formal medical treatment for 4 years after her breast cancer was first diagnosed. She had pain and bleeding that forced her to accept conventional palliative chemotherapy during that time. Unfortunately, she stopped this treatment after 2 cycles as she was unable to tolerate the therapy. The 'before' photo shows that her entire left breast had been ‘digested’ by her cancer. A malignant ulcer had also developed in her affected breast, with bleeding and a putrid discharge. A large clot also caused the area to retract, resulting in an eye-like appearance. The 'after' photo shows her cancer’s complete clinical response within 6 months of beginning atavistic chemotherapy. This also led to noticeable fibrosis and scar retraction in the site.
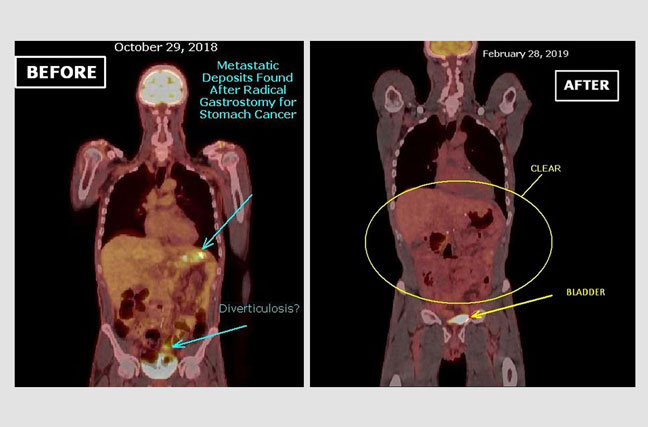
Recurrent Stomach Cancer in the Abdominal Cavity:
This 54 Y/O patient underwent an extensive surgery for a primary cancer of the stomach. Approximately 90% of his stomach was removed, together with a segment of the liver, the entire spleen, gallbladder, and a portion of the colon. However, cancer cells were found by pathology in the margins of the resected stomach. The before photo shows a PET-CT scan image two month after surgery. The image depicts the presence of tumor metabolic activity in the left upper quadrant of the abdomen suggestive of recurrent metastatic deposits. The after photograph represents a PET-CT scan image three months after the initiation of atavistic chemotherapy demonstrating that the increased metabolic activity in the upper left quadrant has disappeared.
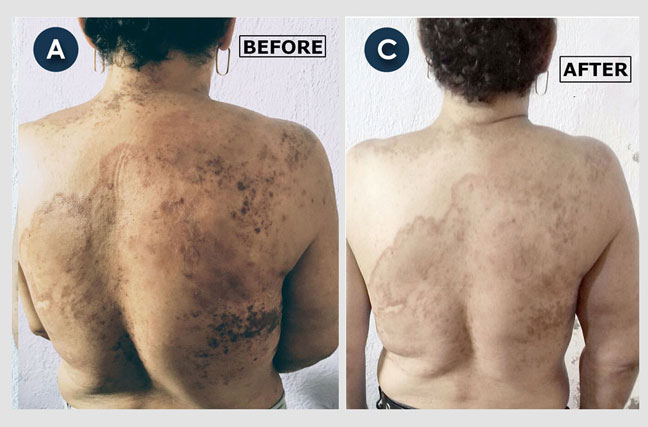
Recurrent Breast Cancer Invading Throughout the Skin of the Patient (part 2):
Gloria, a 43-year-old woman, developed recurrent breast cancer around a surgical scar. For two years, and despite several cycles of traditional chemotherapy, the cancer continued invading and migrating throughout the skin of her chest and abdomen (front and back), causing a constant burning sensation and itching. The ‘before’ photos depict the changes to her abdominal and chest skin. Multiple skin biopsies revealed that the entire area was affected by cancerous cells. The 'after' photo shows Gloria’s amazing progress two months after beginning atavistic chemotherapy.
Recurrent Breast Cancer Invading Throughout the Skin of the Patient: part 1:
Gloria, a 43-year-old woman, developed recurrent breast cancer around a surgical scar. For two years, and despite several cycles of traditional chemotherapy, the cancer continued invading and migrating throughout the skin of her chest and abdomen (front and back), causing a constant burning sensation and itching. The ‘before’ photos depict the changes to her abdominal and chest skin. Multiple skin biopsies revealed that the entire area was affected by cancerous cells. The 'after' photo shows Gloria’s amazing progress two months after beginning atavistic chemotherapy.
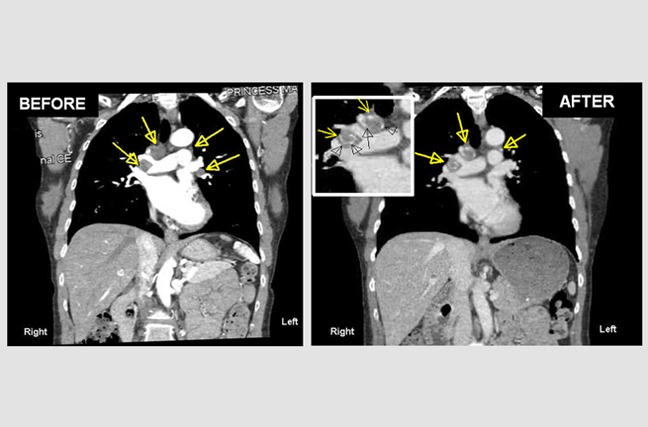
Lymph Node Metastases in the Mediastinum from a Malignant Melanoma.
Alice, a 55-year-old woman, came to us with multiple metastatic tumors in the lymph nodes of the center of her chest (mediastinum). The yellow arrows indicate her melanoma-enlarged lymph nodes. The 'after' photo is one year after she began atavistic chemotherapy, and when the treatment was discontinued. As you can see, lymph nodes are now calcified (hardened) where the melanoma cancer cells have died.
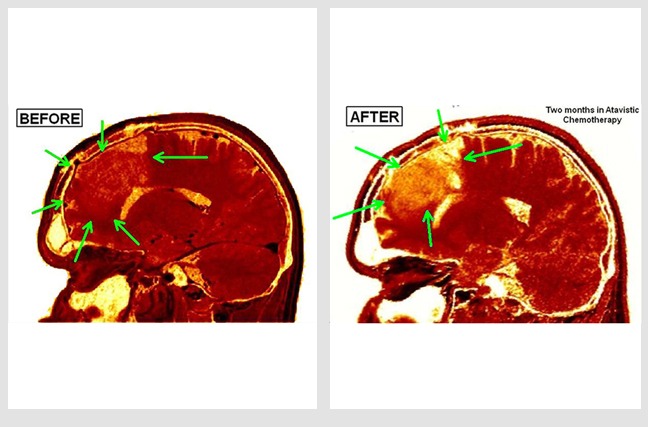
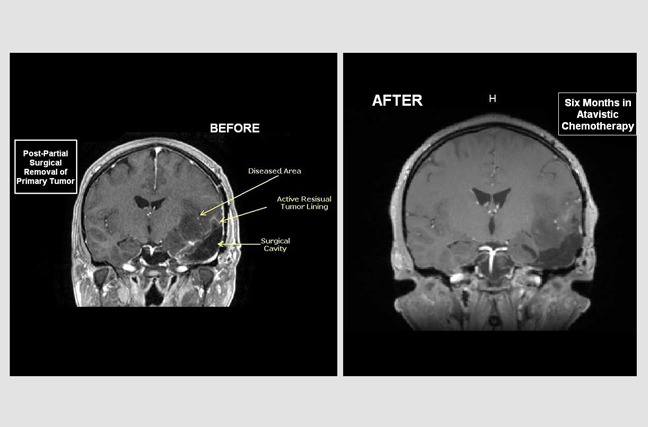
Glioblastoma Multiforme Partially Removed Surgically
Judith is a 32-year-old woman who was diagnosed with an aggressive brain tumor (glioblastoma). The 'before' MRI image was taken around 20 days after an operation to remove approximately 60% of the tumor. The 'after' MRI image follows six months of atavistic chemotherapy. Her treatment was complete in just eight months.
Stage IV, Breast Cancer:
Alejandra is a 45-year-old woman who had been diagnosed with invasive triple-positive breast cancer. She wasn’t able to tolerate any traditional chemotherapy in preparation for a radical mastectomy. As a result, the mastectomy never took place. The 'before' image is 1.5 years after her first diagnosis, and almost a year after abandoning traditional chemotherapy. The 'after' image shows her progress four months after choosing atavistic chemotherapy.
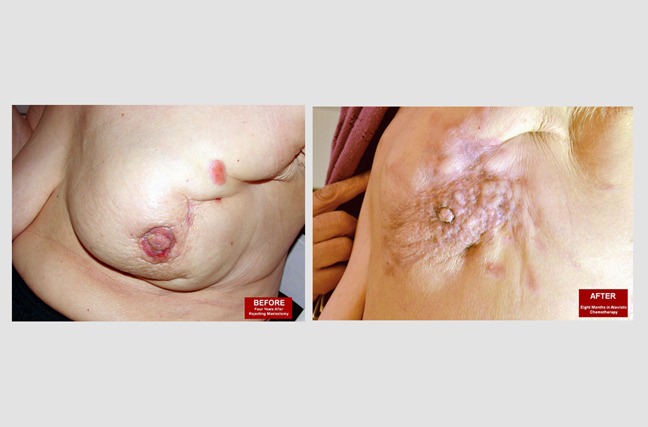
Locally Advanced Breast Cancer:
These pictures show Alice, who is a 75-year-old woman diagnosed with invasive triple-positive breast cancer. The patient did not want to have a radical mastectomy. The 'before' image shows her cancer four years after being diagnosed. The 'after' image was taken eight months after beginning atavistic chemotherapy.
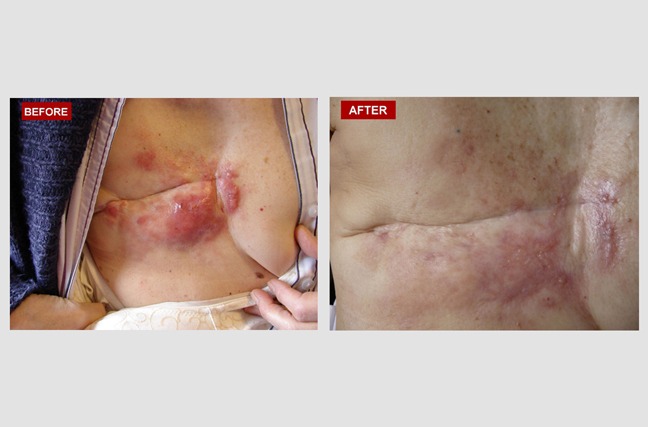
Recurrent Triple-Negative Breast Cancer:
Linda is a 59-year-old woman who had a second recurrence of cancer in the same place. The first recurrence happened 6 months after her original lump was removed. The second recurrence occurred 6 months after she had had a radical mastectomy, traditional chemotherapy and radiation. The 'before' picture shows many raised and hyperemic (red) areas of cancerous tissue growing around her surgical scar. The 'after' image illustrates a dramatic change following four months of atavistic chemotherapy.
Recurrent Breast Cancer at Site of Mastectomy and Distant Metastases:
Susan is a 40-year-old woman whose cancer returned in the same area two years after she had a radical mastectomy, traditional chemotherapy and radiation. The 'before' image shows the extensive ulceration of her skin about six months she was diagnosed. The 'after' image was taken six months after beginning atavistic chemotherapy.
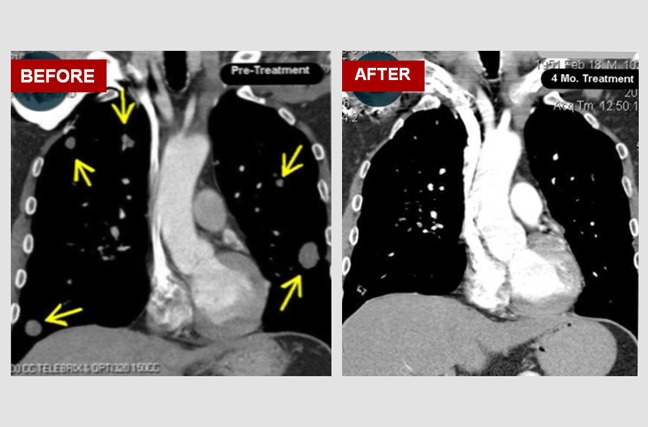
Multiple Lung Metastases from a Colon Cancer:
Terry is a 50-year-old man who came to us with multiple lung metastases (secondary growths) from a colon cancer that had been surgically removed two years earlier. The 'before' image is of a chest CT scan showing large metastatic (secondary) tumors in his lungs (yellow arrows). The 'after' image is a chest CT scan four months after initiating atavistic chemotherapy.
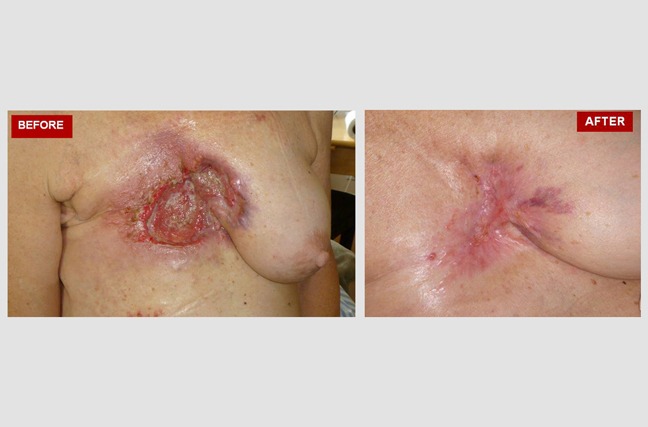
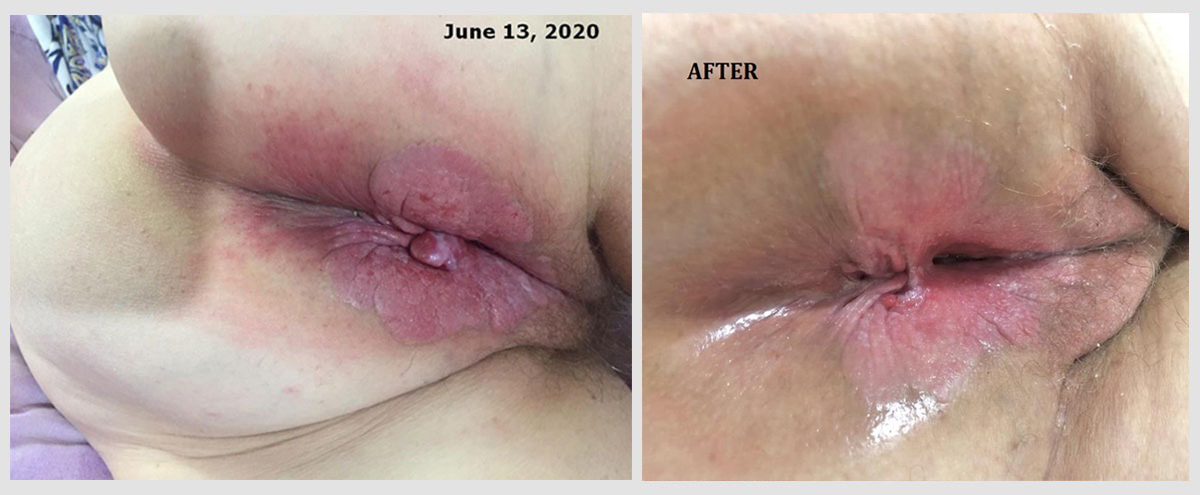
Extramammary Perianal Paget's Disease (rare skin cancer).
This anonymous female patient of 74 years of age, was diagnosed with this rare skin cancer of the perianal and perineal region in June 2020. Different to other skin cancers, this is an adenocarcinoma. The origin of the neoplastic cells could be apocrine glands or epithelial stem cells. Skin areas rich in apocrine glands such as the genital region are typical sites of extramammary Paget’s disease. Surgery was not an option, and the patient rejected radiation and/or traditional chemotherapy. The ‘before’ picture was taken on June 13, 2020. Atavistic chemotherapy was initiated on June 16, 2020. The ‘after’ photo was taken on July 13, 2020 (27 days in atavistic chemotherapy).
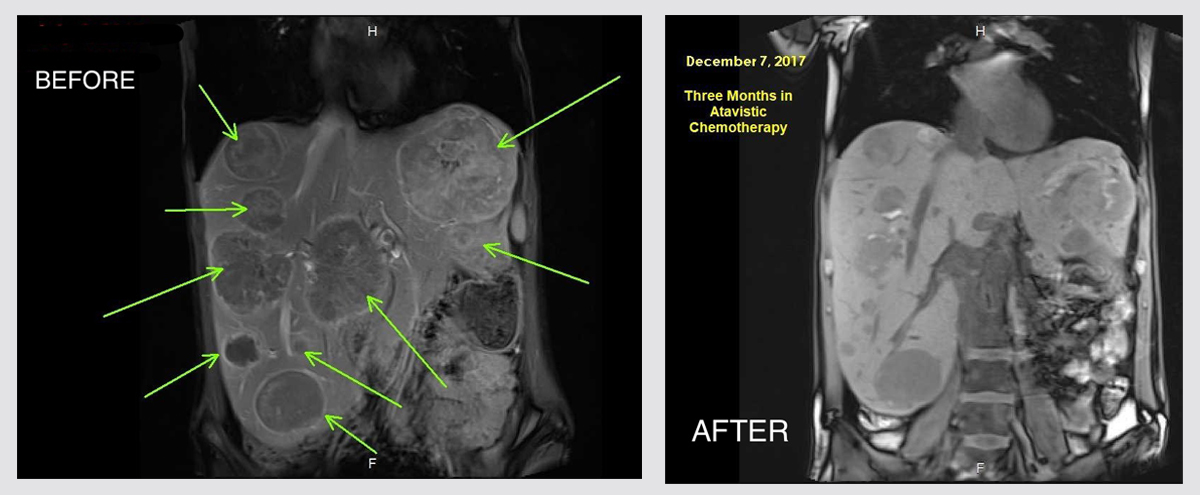
Terminal and Heavily Treated Metastatic Ocular Melanoma with Massive Liver and Bone Metastases
Female patient from the country of Georgia of 34 years of age, who at the age of 31 was diagnosed as suffering of ocular (Uveal/Choroidal) melanoma of the left eye. The affected eye was enucleated and replaced with an eye prosthesis. The patient remained asymptomatic until two years later when liver and bone metastases were discovered. The patient underwent treatment at the Acebadem Clinic in Istanbul, Turkey, where she was treated with KEYTRUDA® (pembrolizumab), and subsequently with GLEEVEC® (Imatinib Mesylate). After a year of continued tumor progression and health deterioration, the patient requested further assistance in Germany at Heidelberg University Hospital where she received Chemo-Saturation (Intra-Hepatic Arterial Infusion of Melphalan). In July 2017, the patient contacted me and asked for my assistance. I was very reluctant to enroll this patient in the atavistic chemotherapy trial, because the amount of cancer in her body was overwhelming and beyond treatment. On the other hand, long periods of treatment with traditional chemotherapy, particularly with alkylating agents such as melphalan, frequently generates multidrug resistance in the surviving cancer cells, which are eventually the ones that predominate and progress in the body. My experience with heavily treated cancers is very disappointing. However, the patient insisted. I also considered the fact that she had three young children and wanted to fight for them. I was unable to save the patient. Not only because she was convinced in the hospital of Georgia no to believe in my treatment and be treated with them. But regardless of that, I knew this was a lost battle. Nevertheless, I am presenting here some images of her radiological studies to show the effectiveness of atavistic chemotherapy in spite of massive amounts of cancer.
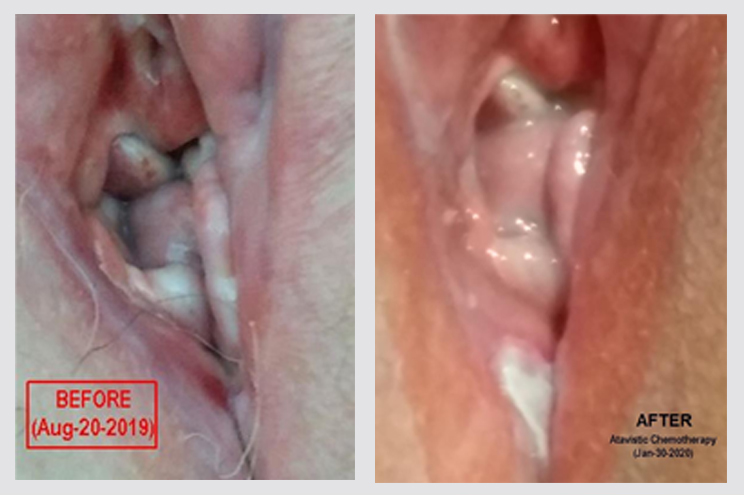
Recurrent Vulval Cancer in a Patient with a History of Vulvar Lichen Sclerosus et Atrophicus
Female patient (57 years old) with vulvar lichen sclerosus et atrophicus for seven years, biopsied in 2012 and 2015, and treated with local steroids (betamethasone). In early 2019, a painful lump in his labia majora of her vulva was biopsied and diagnosed as a well-differentiated squamous cell carcinoma. The bacterial culture of the secretions from the lesion was positive for Staphylococcus aureus, which was treated with antibiotics. For her vulvar malignancy, the patient underwent a partial radical vulvectomy with bilateral sentinel inguinal femoral lymph node dissection and completion lymph node dissection. Ultrasound vulvar and perianal ablation were also carried out. Cancer cells were found invading the surgical margins during a microscopic analysis of the surgical specimen. An acetic acid test three months later suggested a recurrence, which was confirmed by a new biopsy. The patient was offered more surgery, chemotherapy, and radiation, but she declined. In August 2019, she enrolled in our clinical trial. The photographs below where taken by the husband of the patient and show the affected area. However, I do not know from where the biopsy for the recurrence diagnosis was obtained. Nevertheless, after three months of atavistic chemotherapy, the vulvar area (what remains of it) appears healthier than before treatment, and the patient remains cancer-free to this day. The lichen sclerosus is still affecting the patient
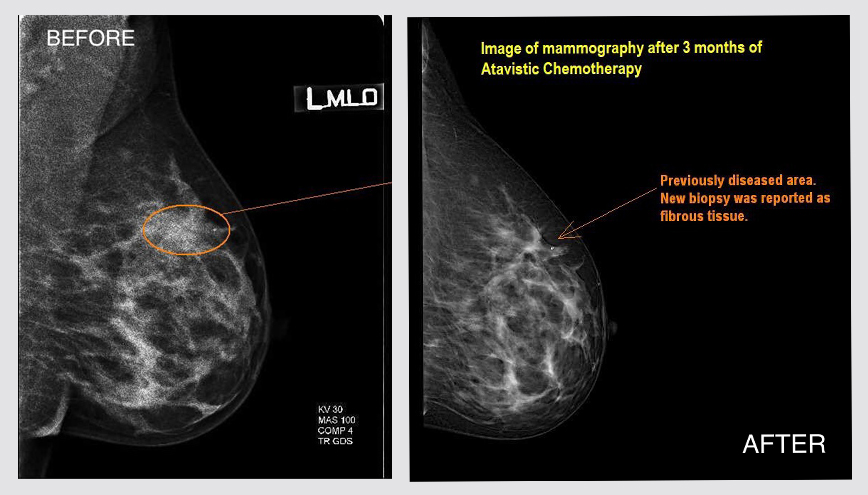
Localized Primary Breast Cancer
Female patient (52 years old) with a mammography and ultrasound describing a worrisome irregular solid region of approximately 15.5 x 7.4 x 10.3 mm and located in the left breast at 3 o'clock, BIRADS 4 (see below). Core Needle Biopsy reveals an invasive ductal carcinoma . Cancer cells are E&P positive and Her-2 negative in immunohistochemistry. The patient desires to avoid a mastectomy and traditional chemotherapy/radiation. The patient is invited to participate in the atavistic chemotherapy trial. After one month of atavistic chemotherapy, a radiologist is unable to suspect malignancy following a new ultrasound obtained from a different radiological facility. The patient was treated for three months, and a mammogram is performed at the fourth month.
This Work is Dedicated to the Loving
Memory of our Mother

If only the love of your young children could have saved you, you certainly would not have died. But our love and tears were not enough. I never understood why God took you so early from our lives, until now. This book would have never been written. Let’s hope this book and the subsequent work derived from these concepts prevent other families from experiencing the pain and sorrows that we went through with your premature death.
Address
U.S Administrative and Enrolment Office:
The Atavistic Chemotherapy Trial
Bethesda Metro Center
Bethesda, Maryland 20814
Call us:
Tel: 301 – 760 – 7777 (Maryland)
Fax: (301) 690 – 9000 (Maryland)
Clinical Trial Sites:
Dr. Frank Arguello (Medical Atavistic Oncology)
Dr. Salvador Belmont (Surgical Atavistic Oncology)
HOSPITAL LOS ANGELES
Av. Campos Eliseos 9371
Cd Juárez, Chih., 32472
Mexico